  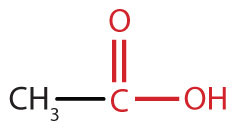
Excellent results are attributed to the decomposition of benzyl nitrite, which is formed by reacting benzyl alcohol and HNO 3 in the presence of multi-purpose GO, by transferring acidic proton from GO to benzyl nitrite. Inspired by the use of HNO 3 for oxidation of alcohol to aldehyde in combination with nanomaterials, we perform alcohol oxidation with GO as Brønsted acidic carbocatalyst and HNO 3 as oxygen carrier leading to extremely high conversion and selectivity using ultra-low carbocatalyst loading (5 wt%). Although this strategy gave a dramatic increase in both reactivity and selectivity, sub-stoichiometric quantities of carbon materials (20–50 wt%) were still required to maximise product yield and selectivity. Recently, the Kakimoto and Peng’s research groups independently attempted to use HNO 3 in combination with nanoshell carbon 23 or carbon nanotubes 10 as promoters for the selective oxidation of primary alcohols to aldehydes. The re-oxidation step was the most retarding process in this study and thus requiring over-stoichiometric amounts of GO for efficient conversions. Reduced graphene oxide (rGO) is formed after dehydration and regenerates to GO under aerobic conditions. It revealed that the epoxy group on GO moieties played a vital role as a hydride acceptor and base for the abstraction of protons from alcohols (Fig. Importantly, a detailed reaction mechanism for the oxidation of alcohol using GO or N-doped graphenes was recently proposed by the Subramanian group, based on density functional theory (DFT) calculations. However, since the exposure of oxygen-containing functional groups on its surface allows GO to behave as an oxidant, it is required in over-stoichiometric quantities (200–800 wt%) in such reactions. In particular, the design of the graphene functionalisation processes helps their utility in a wide range of synthetic transformations (e.g., oxidation of olefins 18, alcohols 19, 20, and sulfides 21, and hydration of alkynes 22). Among them, graphene oxide (GO) is emerging as a new class of heterogeneous catalyst due to its unique property of being easily manipulated with additional functional groups, such as epoxy, hydroxyl, and carboxylic acid, which are arbitrarily distributed in the carbon sheets 16, 17. Recently, carbocatalysts, which incorporate fullerenes 1, 2, 3, nanodiamonds 4, 5, 6, carbon nanotubes 7, 8, 9, 10, 11, and graphite/graphene 12, 13, 14, 15 have attracted great attention as potential sustainable catalysts. The development of green and sustainable chemistry utilising a transition metal-free approach has been connected with the use of non-toxic, robust catalysts with minimal or no adverse environmental or social impact. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
